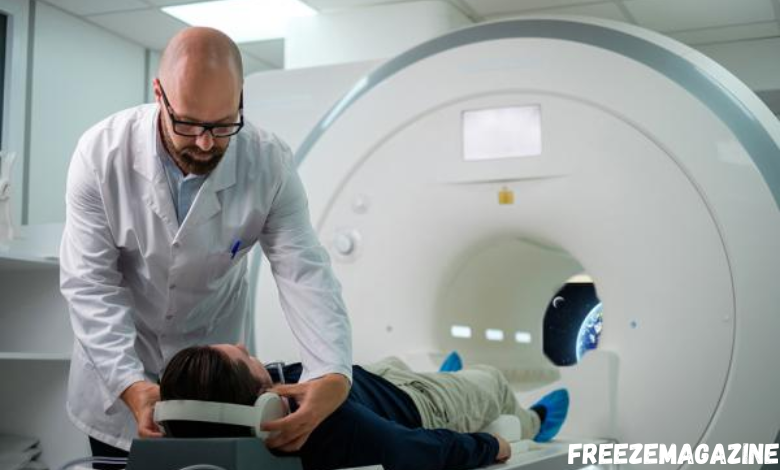
MRI-Safe vs. MRI-Compatible Headphones: The $50,000 Mistake Most Radiology Departments Make
Radiology departments operate under constant pressure to maintain patient safety while managing operational costs and regulatory compliance. When magnetic resonance imaging equipment malfunctions due to improper accessories, the financial impact extends far beyond immediate repair costs. A single incident involving incorrect headphone selection can result in equipment damage, prolonged downtime, rescheduled procedures, and potential regulatory scrutiny that collectively reaches tens of thousands of dollars in losses.
The distinction between MRI-safe and MRI-compatible audio equipment represents one of the most misunderstood aspects of imaging facility management. This confusion stems from similar terminology that masks fundamentally different safety classifications and operational implications. Understanding these differences becomes critical as healthcare facilities face increasing scrutiny over equipment protocols and patient safety standards.
Many radiology administrators assume that any headphones marketed for medical use will function appropriately in MRI environments. This assumption has led to equipment failures, safety incidents, and costly operational disruptions that could have been prevented through proper classification understanding and procurement processes.
Understanding MRI Safety Classifications in Audio Equipment
MRI safety classifications follow strict definitions established by the American Society for Testing and Materials, with each category representing specific levels of magnetic field interaction and patient risk.
Hospital headphones fall into one of three distinct safety categories, each with different implications for patient safety and equipment protection. The classification system exists because magnetic fields in MRI environments can cause ferromagnetic objects to become projectiles, generate heat in conductive materials, or interfere with imaging quality through electromagnetic disturbance.
MRI-safe devices contain no metal components and pose no known hazards in any MRI environment. These devices will not be attracted to the magnetic field, will not heat up during scanning, and will not interfere with image quality. The designation applies regardless of magnetic field strength or specific MRI scanner configuration.
MRI-compatible devices may contain some metal components but have been tested to demonstrate acceptable safety and functionality in specific MRI environments. However, their use requires verification of compatibility with particular scanner specifications, field strengths, and operational parameters before implementation in clinical settings.
MRI-unsafe devices contain ferromagnetic materials or electronic components that pose definite hazards in MRI environments. These devices can become projectiles, cause burns through heating, or significantly degrade image quality through electromagnetic interference.
The Hidden Costs of Classification Confusion
Equipment damage represents the most visible cost when departments mistake MRI-compatible devices for MRI-safe alternatives. Ferromagnetic headphones can damage scanner components, requiring manufacturer service calls that typically range from several thousand to tens of thousands of dollars depending on the extent of magnetic field interaction and resulting mechanical stress.
Operational downtime compounds the initial equipment damage through lost productivity and rescheduled procedures. MRI scanners generate revenue through continuous utilization, and unexpected downtime disrupts carefully managed scheduling systems that often book procedures weeks in advance. Each day of scanner downtime can represent significant lost revenue while fixed operational costs continue.
Regulatory and Liability Implications
Safety incidents involving improper MRI accessories trigger regulatory reporting requirements and potential investigations by oversight bodies. The Joint Commission and other accrediting organizations scrutinize MRI safety protocols, and documented failures can result in compliance findings that require expensive remediation efforts and ongoing monitoring.
Patient safety incidents create liability exposure that extends beyond immediate medical concerns. Even minor injuries caused by heating or projectile effects can result in legal action, insurance claims, and reputation damage that affects long-term patient volume and referral patterns.
Staff Training and Protocol Development Costs
Incidents involving incorrect headphone classification often reveal broader gaps in staff training and equipment protocols. Addressing these gaps requires comprehensive retraining programs, updated procedures, and enhanced oversight systems that consume significant administrative resources and staff time.
The complexity of managing multiple headphone types with different safety classifications increases training requirements and creates ongoing confusion among technologists who must make real-time decisions about equipment use during patient procedures.
Operational Decision Points for Equipment Selection
Patient comfort requirements drive many headphone selection decisions, but these needs must be balanced against safety classifications and operational constraints. MRI procedures often involve lengthy scan times where patient anxiety and claustrophobia can compromise image quality through movement or premature procedure termination.
Audio quality expectations vary significantly between diagnostic imaging requirements and patient comfort applications. Some procedures require precise audio communication between technologists and patients, while others focus primarily on noise reduction and anxiety management through music or ambient sounds.
Integration with Existing Systems
Many radiology departments operate mixed equipment environments with scanners from different manufacturers and varying field strengths. This complexity requires headphone solutions that maintain consistent safety classifications across all operational scenarios rather than requiring different equipment for different scanners.
Inventory management becomes more complex when departments maintain separate headphone stocks for different safety requirements. Staff must track, maintain, and properly deploy multiple equipment types while ensuring that safety classifications match specific use cases and scanner environments.
Maintenance and Replacement Considerations
MRI-safe headphones typically require less complex maintenance protocols because their non-metallic construction eliminates concerns about component degradation that could affect magnetic properties. This simplicity reduces long-term operational complexity and staff training requirements.
Replacement scheduling becomes more predictable when equipment classifications remain constant throughout the device lifecycle. MRI-compatible devices may require periodic retesting or recertification to verify continued safety performance, while MRI-safe alternatives maintain their classification regardless of wear or component aging.
Risk Mitigation Through Proper Classification
Standardizing on MRI-safe audio equipment eliminates classification-related decision points during routine operations. Staff can focus on patient care rather than evaluating equipment compatibility for specific scanners or procedures, reducing the cognitive burden during busy operational periods.
This standardization approach proves particularly valuable during emergency procedures or when temporary staff members may be unfamiliar with specific equipment protocols. Clear, consistent safety classifications reduce the likelihood of errors during high-pressure situations where comprehensive equipment evaluation may be impractical.
According to the FDA’s guidance on MRI safety, proper equipment classification and staff training represent critical components of comprehensive magnetic resonance safety programs that protect both patients and healthcare workers.
Quality Assurance Integration
Incorporating headphone safety verification into routine quality assurance programs ensures ongoing compliance with safety protocols and identifies potential issues before they result in operational problems. Regular equipment audits can verify that only appropriately classified devices remain in active use.
Documentation requirements for MRI safety programs typically include equipment inventories, safety classifications, and staff training records. Maintaining accurate records becomes simpler when equipment classifications remain consistent and clearly defined throughout the facility.
Long-term Strategic Considerations
Equipment procurement decisions made today will influence operational safety and costs for years to come. Selecting MRI-safe alternatives eliminates future compatibility concerns when upgrading scanners or expanding imaging capabilities, providing greater flexibility for long-term strategic planning.
Budget planning becomes more predictable when equipment classifications eliminate variables related to safety compliance and compatibility verification. Departments can focus financial resources on patient care improvements rather than managing complex equipment compatibility matrices.
Technology Evolution and Future-Proofing
MRI technology continues advancing with higher field strengths and improved imaging capabilities that may affect compatibility requirements for existing equipment. MRI-safe devices provide greater assurance of continued usability regardless of future scanner upgrades or replacement decisions.
Emerging imaging protocols and specialized procedures may introduce new operational requirements that benefit from simplified equipment management. Having consistently safe audio equipment removes one variable from the complex process of implementing new clinical capabilities.
Conclusion
The distinction between MRI-safe and MRI-compatible headphones extends far beyond technical specifications to encompass patient safety, operational efficiency, and long-term cost management. Understanding these differences enables radiology departments to make informed procurement decisions that protect both immediate operational needs and future strategic flexibility.
The $50,000 mistake referenced in this analysis reflects not just equipment damage costs, but the cumulative impact of downtime, regulatory compliance, staff retraining, and ongoing operational complexity that results from improper equipment classification. These costs can be avoided through careful attention to safety classifications and their operational implications.
Successful MRI safety programs require clear equipment standards, comprehensive staff training, and consistent implementation of classification protocols. By prioritizing MRI-safe alternatives where operationally feasible, departments can reduce risk exposure while maintaining the patient comfort and clinical functionality that drive positive imaging outcomes.




Post Comment